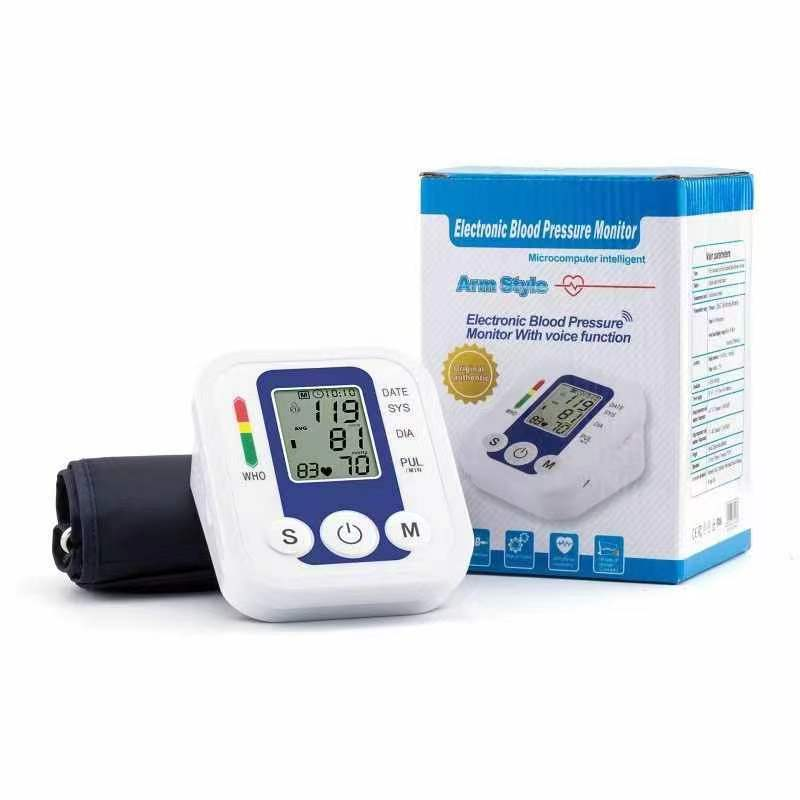
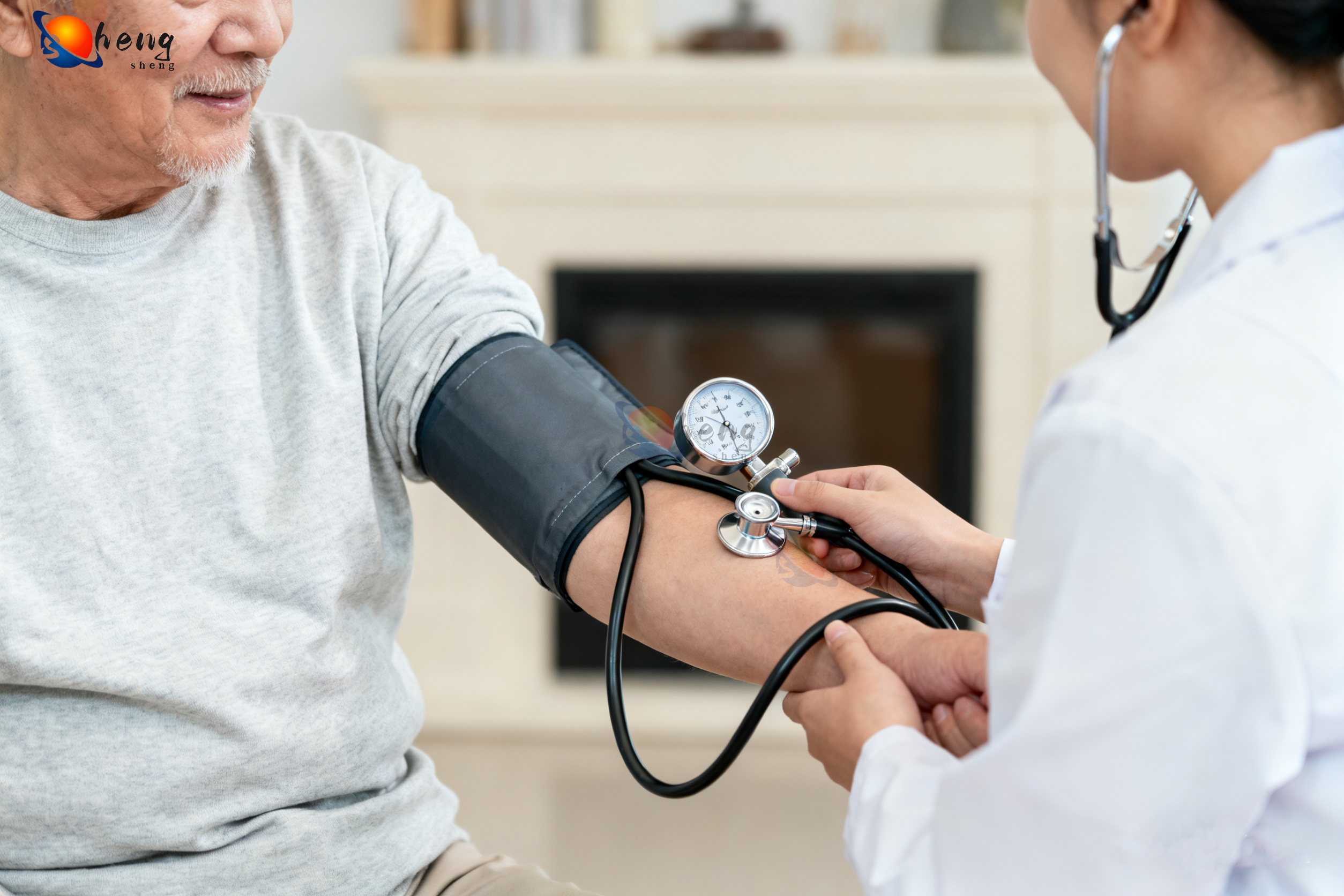
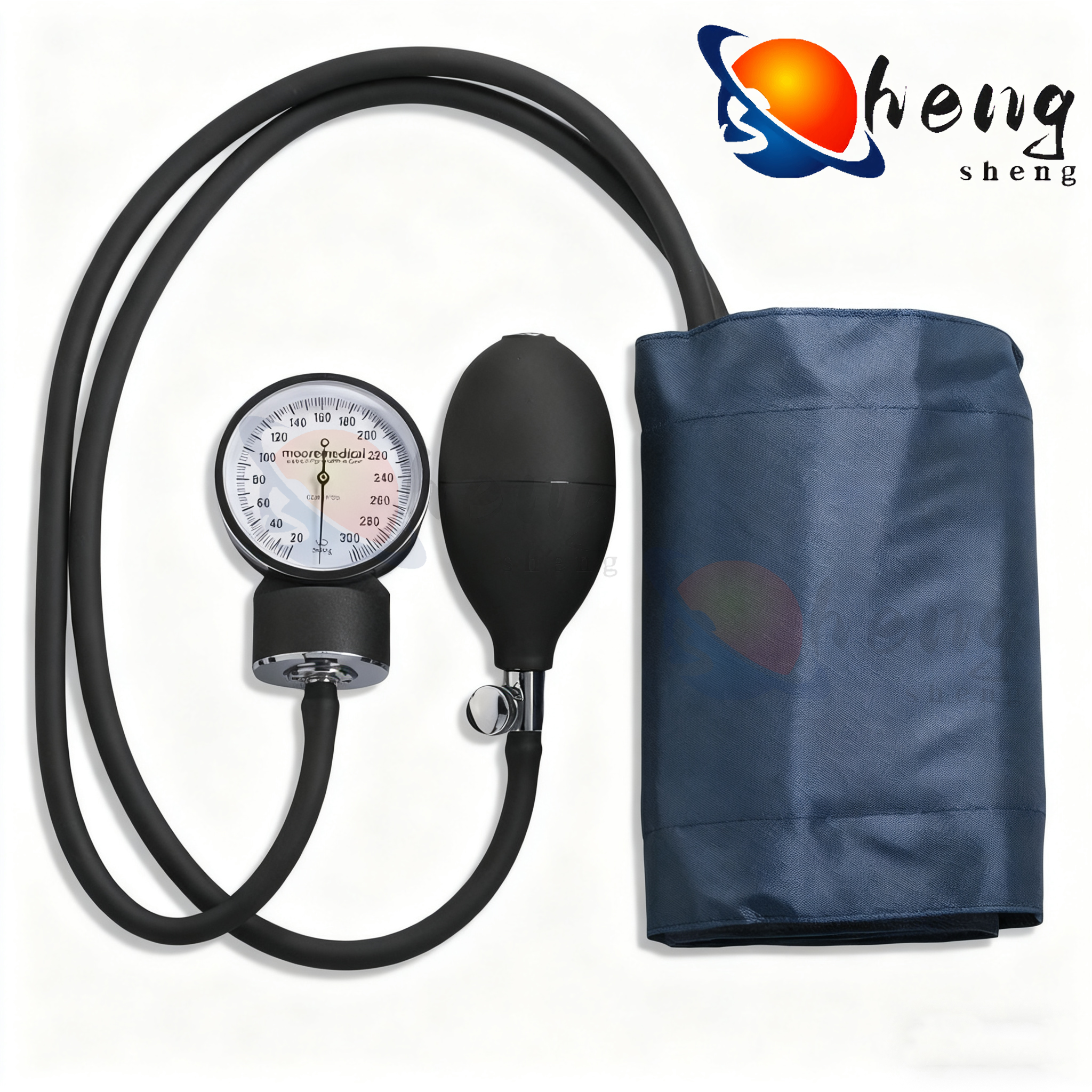
Professional-Grade Reliability and Connectivity
The high quality blood pressure cuff delivers professional-grade reliability through robust engineering standards that ensure consistent performance under demanding clinical conditions and extended operational periods. This reliability foundation begins with comprehensive quality control testing that subjects each device to thousands of inflation cycles, temperature extremes, and mechanical stress evaluations before clinical deployment. The high quality blood pressure cuff utilizes premium electronic components with extended operational lifespans, reducing the likelihood of measurement failures during critical patient care situations. Advanced connectivity features enable seamless integration with modern healthcare technology ecosystems, supporting telemedicine initiatives and electronic health record management systems. Bluetooth wireless technology allows automatic data transmission to smartphones, tablets, and computer systems, eliminating manual data entry errors and streamlining patient monitoring workflows. The high quality blood pressure cuff supports multiple user profiles with individual memory storage capabilities, accommodating shared use scenarios in clinical settings and multi-patient home care environments. Professional software applications provide comprehensive data analysis tools including trend graphing, statistical summaries, and customizable reporting features that support clinical decision-making processes. The device maintains measurement accuracy across wide temperature and humidity ranges, ensuring reliable operation in various environmental conditions from air-conditioned hospitals to outdoor mobile health units. Battery life optimization provides extended operational periods between charging cycles, supporting continuous monitoring protocols without interruption concerns. The high quality blood pressure cuff features automatic power management systems that extend battery life while maintaining measurement precision and connectivity capabilities. Regulatory compliance encompasses FDA approval, CE marking, and international medical device certifications that validate performance standards and safety requirements. Built-in diagnostic systems continuously monitor device performance parameters, providing early warning indicators for maintenance needs or calibration requirements. The professional-grade construction includes sealed electronic components that resist moisture infiltration and cleaning chemical exposure common in clinical environments. Service support networks provide technical assistance, calibration verification services, and replacement part availability that ensure long-term operational reliability for healthcare facilities and individual users investing in the high quality blood pressure cuff technology.